
Phenol is also a versatile precursor to a large collection of drugs, most notably aspirin but also many herbicides and pharmaceutical drugs. 
Nonionic detergents are produced by alkylation of phenol to give the alkylphenols, e.g., nonylphenol, which are then subjected to ethoxylation. Partial hydrogenation of phenol gives cyclohexanone, a precursor to nylon. 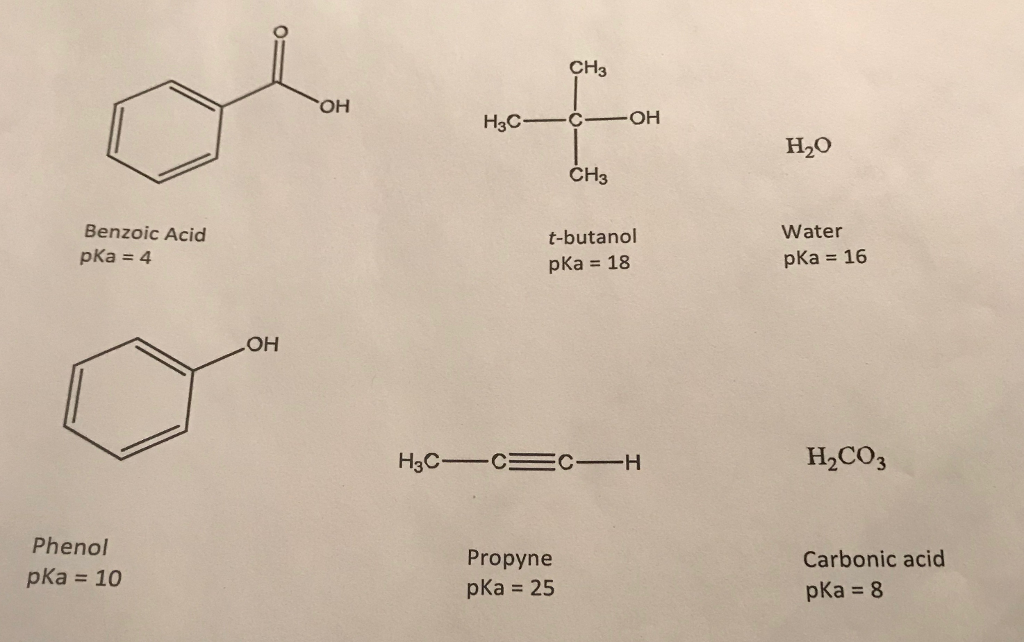
Condensation of phenol, alkylphenols, or diphenols with formaldehyde gives phenolic resins, a famous example of which is Bakelite. Condensation with acetone gives bisphenol-A, a key precursor to polycarbonates and epoxide resins. The major uses of phenol, consuming two thirds of its production, involve its conversion to precursors for plastics. 8 - 12 it is in equilibrium with the phenolate anion C 6H 5O − (also called phenoxide): C 6 H 5 OH ↽ − − ⇀ C 6 H 5 O − + H + The sodium salt of phenol, sodium phenoxide, is far more water-soluble. Homogeneous mixtures of phenol and water at phenol to water mass ratios of ~2.6 and higher are possible. Phenol is an organic compound appreciably soluble in water, with about 84.2 g dissolving in 1000 mL (0.895 M). 2.3.1 Hydrolysis of benzenesulfonic acid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
